
We see them at almost every festivity, during New Year's Eve, 4th of July and even at amusement parks. But have you ever wondered how this amazing colored light show takes place? Well, it is all about chemistry.
First of all, fireworks were first produced in China by filling a tubular piece with gunpowder. Gunpowder is an explosive that usually consists of a mixture of sulfur, charcoal and potassium nitrate. This chemical mixture along with produced a loud noise that was not used for entertainment more than 1,000 years ago. Nowadays, after popular demand, this same mixture is used to create an amazing colorful scenery in the night sky. The early explosives that were first created did not involve color, but later they experimented with charcoal and steel which resulted in yellow and orange tones.
But, what other metals combine to produce the rest of the colors? What reactions do these metals carry out to emit the colors? The color red is produced by strontium compounds, green is produced by barium and cooper produces blue. The color's emission takes place after heating these metals. Thus the atoms of each element absorb the energy released by the heat reaction and discharge it in the form light. Depending on the amount of energy, is the color emitted. The higher the energy, the shorter the wavelength. The lower the energy, the longer the wavelength. This can be observed in the following wavelength diagram.
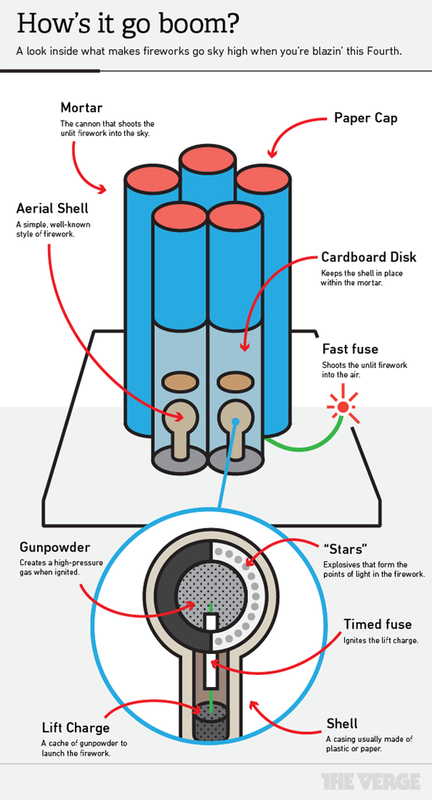
So now we understand how the colors are transmitted by this spectacle. But, how are fireworks assembled to give us such organized and synchronized patterns of light? As I mentioned before, the materials used for fireworks are put together in a tube that is in charge of shooting the unlit firework into the sky. Inside that tube is a shell which is what actually flies up to the sky and produces the light and color effects. The shell itself consists of a certain packaging pattern which includes gunpowder, stars (these are the lighted spots we see in the sky when the firework lights up), a lift charge (which launches the firework) and a fuse that is timed to create the explosion once the shell has reached the sky. The following diagram describes this structure perfectly.
First of all, fireworks were first produced in China by filling a tubular piece with gunpowder. Gunpowder is an explosive that usually consists of a mixture of sulfur, charcoal and potassium nitrate. This chemical mixture along with produced a loud noise that was not used for entertainment more than 1,000 years ago. Nowadays, after popular demand, this same mixture is used to create an amazing colorful scenery in the night sky. The early explosives that were first created did not involve color, but later they experimented with charcoal and steel which resulted in yellow and orange tones.
But, what other metals combine to produce the rest of the colors? What reactions do these metals carry out to emit the colors? The color red is produced by strontium compounds, green is produced by barium and cooper produces blue. The color's emission takes place after heating these metals. Thus the atoms of each element absorb the energy released by the heat reaction and discharge it in the form light. Depending on the amount of energy, is the color emitted. The higher the energy, the shorter the wavelength. The lower the energy, the longer the wavelength. This can be observed in the following wavelength diagram.
So now we understand how the colors are transmitted by this spectacle. But, how are fireworks assembled to give us such organized and synchronized patterns of light? As I mentioned before, the materials used for fireworks are put together in a tube that is in charge of shooting the unlit firework into the sky. Inside that tube is a shell which is what actually flies up to the sky and produces the light and color effects. The shell itself consists of a certain packaging pattern which includes gunpowder, stars (these are the lighted spots we see in the sky when the firework lights up), a lift charge (which launches the firework) and a fuse that is timed to create the explosion once the shell has reached the sky. The following diagram describes this structure perfectly.
Now, during the holidays that are coming, while everyone else wonders how it all takes place, you will be able to picture all this information. However, please keep in mind fireworks are dangerous explosives which should only be handled by responsible adults. Enjoy the holidays safely and admire this phenomenal show from afar!
References:

Author: Yaniris Alvarado Luna
Undergraduate Biological Sciences student at the University of Puerto Rico in Aguadilla. Aspiring Veterinarian.
Undergraduate Biological Sciences student at the University of Puerto Rico in Aguadilla. Aspiring Veterinarian.

 RSS Feed
RSS Feed
